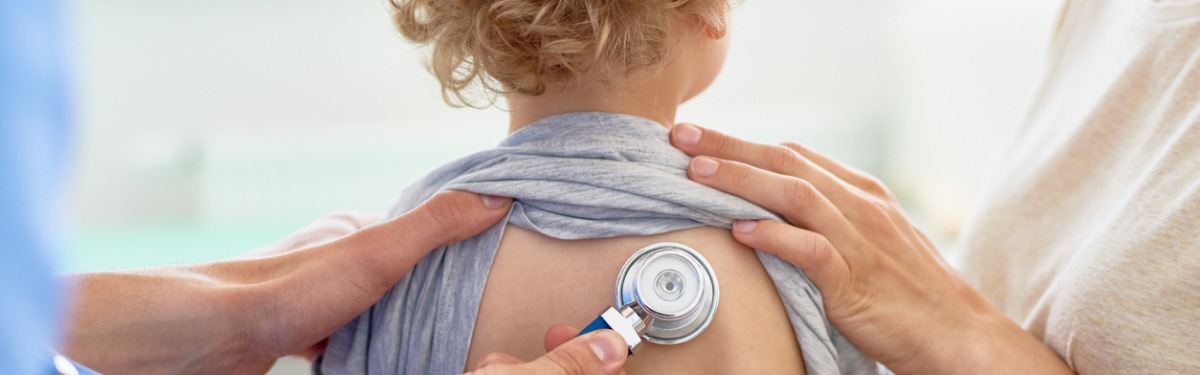
A quick bedside test can deliver diagnosis of respiratory virus infection in children and allow clinicians to treat patients while protecting others from infection. Results from recent trials could change care pathways in the future.
Published: 17 August 2020
Find out how we support medical technology, device and diagnostic companies
Seasonal respiratory infections
Each winter in the UK, paediatric clinicians are faced with a surge in admissions to hospital, most commonly because of respiratory syncytial virus (RSV) infections in infants. While waiting around 24 hours for test results to confirm or rule out the diagnosis, children will often require isolation to prevent infecting others on a ward, and medics will need to wear personal protective equipment.
Parents and clinicians would benefit greatly from rapid diagnosis of RSV infections. Discussions about next steps would be clearer, confirmation means a reduction in other potentially more invasive tests, and knowing whether or not a child has a viral infection has an impact on control measures. In the next few years, medications for RSV infections may become available that are likely to work more effectively early on in an illness and be expensive - another reason for a quick accurate answer.
Researchers at the NIHR Newcastle In Vitro Diagnostics Co-operative (MIC) investigated a small and rapid test that can be used at the bedside to provide a result in around 20 minutes, to see if it could improve diagnosis of RSV infections.
Dr Joy Allen, a senior methodologist at the NIHR Newcastle MIC, says: “Our remit at the MIC is to evaluate new diagnostics to understand what the care benefits may be for patients in the NHS. We generate evidence specific for UK adoption with the aim of getting new diagnostics that will lead to benefit into the NHS quicker.
She continues: “In this case, we were predominantly interested in how a rapid, bedside test for RSV compared with standard laboratory testing and whether it gave as accurate a result. But we were also interested in what the acceleration of results could mean for patient management.”
Trialling a rapid diagnostic
Along with Dr Malcolm Brodlie, a clinical academic in paediatric respiratory at Newcastle Hospitals and Newcastle University, Dr Allen and colleagues conducted a clinical evaluation of the new diagnostic test across two hospitals: the Great North Children’s Hospital, with an on-site laboratory, and the Sunderland Royal Hospital, which relied on an offsite service with a longer return time. The device was produced by Roche, but the study was entirely investigator led.
The team began with a small pilot during the 2016/17 winter season, to ensure that their procedures were correct and that they could hit the ground running the following year, recruiting enough patients to adequately run the study.
The full study in 2017/18 involved 186 patients with an average age of five and a half months, whose families gave consent to their participation. Samples were taken of the children’s respiratory secretions - nasal mucus - and split to be sent for standard testing as well as run through the new bedside test. Once the standard laboratory test result was available, patients were treated in the appropriate way.
As an observational study, only the standard test results were clinically acted on, but both results were recorded. Data were also recorded on how long the patient was in hospital, the isolation procedures used and what other tests and medicines they received.
The bedside test was found to have high diagnostic accuracy, and the time to result was an average of 36 minutes compared to an average of almost 29 hours for the laboratory test. The team also found potential for cost savings from adopting the smaller more rapid test.
The NIHR Clinical Research Network (CRN) supported the research study by assisting with the recruitment of participants.
Read more making a difference stories.
Collaborating to achieve success
Dr Brodlie says: “A large volume paediatric study with this number of recruits is relatively rare, and this was genuinely a fantastic collaboration between the university, the NIHR MIC team, the two hospitals and industry. We are very pleased too that right from the start there were families involved - they were able to tell us what they thought was important and the way they would like this to look.”
While designing the study, the team involved a focus group with parents to discuss procedures, and during the course of their testing interviews took place with parents of children involved as well as clinicians who work in the area. Parents wanted their child to receive support for their symptoms, and a rapid diagnosis was seen as a real benefit in that regard. "I think as a parent it gives you peace of mind knowing actually what's wrong" and "As a parent, you want to know what's wrong with your child" are two examples of comments that arose.
Parents also expressed concern for other patients in the hospital: limiting their exposure to a virus which may make them sicker is of utmost importance. One said: ‘’You don’t know what a child’s immune system is like and you could be putting them in danger. If somebody else has that, they could be putting XXX in danger because [their] immune system is as weak as it is.’’
Clinicians appreciated the result as an aid to discussions with parents about what they were going to do and why. Dr Allen says: “Our study was set in the real world and pragmatic - on one site there were research nurses but on the other site clinical nurses so we got feedback from both about using the test in a real clinical scenario. They believed that it was easy to use and it seamlessly was incorporated into their work.”
Dr Brodlie adds: “As well as the main finding, another really valuable part of the study was the wider modelling of the impact on patient pathways and potential economic impact of this sort of test.” The economic analysis predicted a cost saving of £62 per patient if the test was used.


