Published: 05 July 2020
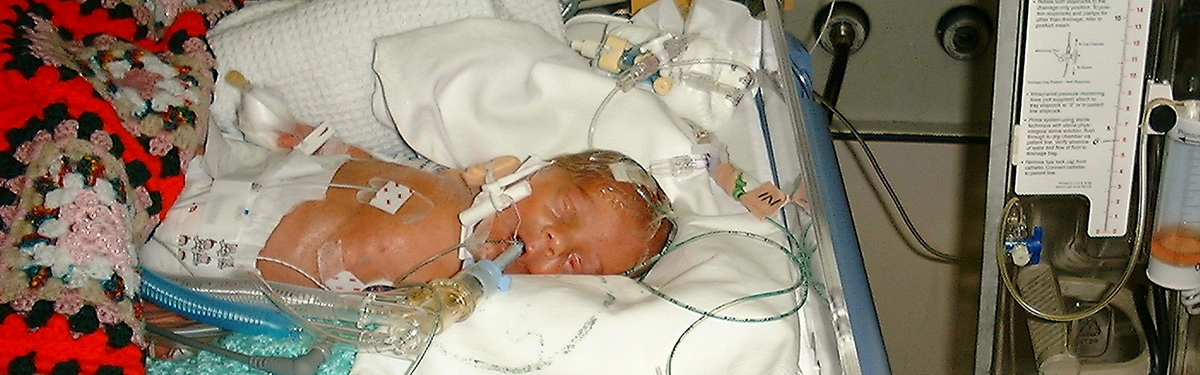
Premature babies with serious brain haemorrhage treated with a novel surgical technique were twice as likely to survive without severe learning disability 10 years later, when compared with infants given standard treatment, NIHR-funded research finds.
The procedure, called ‘Drainage, Irrigation and Fibrinolytic Therapy’ (DRIFT), is the first and only treatment to objectively benefit infants with serious brain haemorrhage, known as intraventricular haemorrhage (IVH) which can lead to severe learning impairment and cerebral palsy.
Findings of the DRIFT10 study are now published in the journal Archives of Diseases in Childhood.
DRIFT aims to reduce disability in premature babies with serious brain haemorrhage by washing out the ventricles in the brain to remove toxic fluid and reduce pressure.
During the ten-year follow-up study, researchers assessed 52 of the 65 survivors from a cohort of 77 premature babies with severe brain haemorrhage who had been recruited for the original DRIFT trial. That trial, funded by Cerebra and the James and Grace Anderson Trust, had run from 2003-2006 and was led by Andrew Whitelaw, Professor of Neonatal Medicine at the University of Bristol and Ian People, Consultant Neurosurgeon from University Hospitals Bristol and Weston NHS Foundation Trust.
On that trial, 39 babies received the DRIFT intervention, and 38 received standard treatment which uses lumbar punctures to control expansion of the ventricles and reduce pressure. An initial two-year follow-up study had shown reduced rates of severe cognitive disability, but researchers wanted to assess whether the DRIFT intervention had longer-term benefits and how it affected their schooling.
In the latest study, Dr Karen Luyt from Bristol Medical School, traced and assessed the children aged ten, and at school, to investigate whether the treatment had led to reduced neurodisability rates.
Using results from cognitive, vision, movement and behaviour assessments, parent and guardian interviews, and educational attainment scores, the team found that the pre-term babies who received DRIFT were almost twice as likely to survive without severe cognitive disability than those who had received standard treatment. They also found that infants given the DRIFT treatment were also more likely to attend mainstream education.
Dr Luyt, a Reader in Neonatal Medicine, said: “Bleeding in the brain is one of the most serious complications of preterm birth and premature babies are particularly at risk of bleeding, the condition can cause significant brain injury leading to subsequent severe learning disabilities.
“The results of this study clearly demonstrate that secondary severe brain injury is reduced in preterm infants by using this neonatal intervention, and importantly, this is sustained into middle school-age.
“We hope that these results will be used to inform UK and international healthcare guidelines and support implementation of DRIFT as a clinical service to help improve outcomes for vulnerable babies.
“We would also like to thank the families and the children who took part in the study for their support and significant contribution that has helped advance our treatment of this condition.”
Dr William van’t Hoff, Chief Executive Officer of the NIHR’s Clinical Research Network (CRN) which supported the study, said: “These landmark results provide the first long-term evidence that this novel intervention can help reduce cognitive disability in infants born with serious brain haemorrhage.
“Central to this trial has been patient and public involvement – one of the NIHR’s key values - with patients and their parents involved throughout the trial design and process, which has led to its success.”
The study was funded by the NIHR’s Health Technology Assessment (HTA) Programme.
More information, including a series of short films explaining the findings, are available on the NIHR Funding Awards website.

