Published: 15 June 2020
A new trial is rolling out across the UK, looking to identify safe and effective alternatives to treatment using a ventilator.
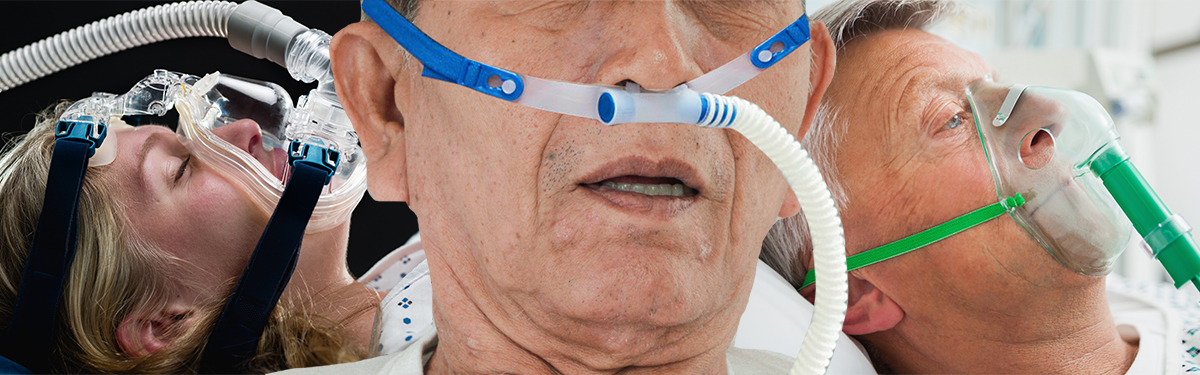
Patients with severe symptoms of COVID-19 who may be struggling to breathe may require respiratory support to help get more oxygen into their lungs. Currently, patients in hospital may be treated with a ventilator in intensive care units or with a less invasive method of respiratory support.
By identifying which non-invasive treatments are most effective, and using these at an earlier stage, it may be possible to reduce the need for invasive mechanical ventilation, and therefore keep more patients out of critical care wards.
The RECOVERY-RS trial (Randomised Evaluation of COVID-19 Therapy - Respiratory Support) aims to compare three existing non-invasive treatments to determine which provides the best outcome for patients. These are: (1) Continuous Positive Airway Pressure (CPAP); (2) High Nasal Flow Oxygen (HFNO); (3) oxygen therapy only.
These treatments are already available in the NHS and have been used for patients with COVID-19, preventing them from needing intensive care services. However, it is not known which is the most effective in the long term. In fact, recommendations for non-invasive treatments for COVID-19 patients across the globe are inconsistent.
High quality evidence is therefore urgently needed to inform clinical practice and to ensure the best long term outcomes for patients. The RECOVERY-RS trial aims to provide that evidence.
The trial is funded and supported by the NIHR, and is one of a number of COVID-19 studies that are considered to be urgent public health research by the UK’s Chief Medical Officers. It has been given priority status by the Department of Health and Social Care through the NIHR’s national prioritisation process. Led by the University of Warwick and Queen’s University Belfast, the trial is also endorsed by both the British Thoracic Society and the Intensive Care Society.
Professor Danny McAuley, Co-Chief Investigator, based at Queen’s University Belfast said:
“The unfolding COVID-19 pandemic brings an urgent need for new therapies, particularly for critically ill COVID-19 patients. We simply do not know what treatments are effective for this new disease and we urgently need this trial to test what is the best way to deliver respiratory support as an alternative to ventilators.”
Professor Gavin Perkins, Co-Chief Investigator based at the University of Warwick Clinical Trials Unit said:
“Clinical trials are the only way to safely study these interventions and to offer patients the additional protections necessary within the carefully controlled environment of a clinical trial, and are the best way to quickly find effective supportive interventions for this global pandemic.”
Video transcript: The RECOVERY-RS trial
Professor Paul Dark, NIHR National Specialty Lead for Critical Care:
"The RECOVERY-RS Urgent Public Health trial addresses a critically important clinical question about the respiratory support of patients with highly suspected severe COVID-19. We have built national networks across the UK's four nations to deliver the aims of the trial over the coming months. I urge all acute NHS hospitals to set up trial delivery immediately so that we can develop definitive evidence for the best acute respiratory care for our patients."
Around 60 hospitals are already taking part in the trial, and more than 120 patients enrolled, but in order to ensure evidence is robust, many more sites and participants are needed. Mindful of the current burden on NHS teams, the study has been designed to be as flexible as possible. For example not all the interventions must be delivered at every site, training is available online, data collection has been minimised, patients may be recruited from any part of the hospital, and it is also possible to co-enrol with other COVID-19 studies.
The research team and NIHR Clinical Research Network can support any hospitals that may want to set up the study.
Visit the trial website

