NIHR Programme Grants for Applied Research (PGfAR) is seeking to increase the diversity of study type and design submitted for funding. This case study presents a research design to assess the application of rapid-learning methodology to real-world data to determine a heart limit dose for lung cancer patients undergoing radiotherapy.
Programme details
Programme name: Using real world data and rapid learning to drive improvements in lung cancer survival
Lead researchers: Professor Corinne Faivre-Finn and Dr Gareth Price
Institution: The Christie NHS Foundation Trust
Funding: £2,376,526
Patient and clinical need
Lung cancer is the leading cause of cancer mortality in the UK. It is mainly treated by radiotherapy, one of the most important and cost-effective interventions. However, it is well-known that radiotherapy can lead to harmful side effects and must be continuously monitored and modified to increase patient benefit.
This research aims to improve current approaches by embedding the Learning Healthcare System concept in routine radiotherapy practice to establish a rapid-learning methodology for the timely, safe and evidence-based evaluation of changes. By using sequential rapid-learning cycles, one can use the effects observed in each cycle to refine the dose limit for use in the next, thereby optimising it for achieving the optimum clinical outcome (for instance, survival vs. severe lung toxicity incidence in this study). Rapid-learning methodologies use real-world data (observational data) routinely collected by clinicians as part of patients’ normal care. Therefore, it is anticipated that rapid learning will enable such improvements to be made as part of routine clinical care. This is likely to embed within the NHS long-term plan of digital transformation, leading to significant service improvement.
Aims of the programme
This study aims to:
- Use rapid learning to introduce a heart dose limit for lung cancer radiotherapy and optimise the limit to get the best clinical outcome
- Determine the challenges and opportunities associated with the use of rapid learning for the evidence-based introduction of changes in radiotherapy practice within the NHS.
Programme design
The rapid-learning methodology represents an alternative to conventional clinical trials, which are challenging for the evaluation of rapid and ongoing technical radiotherapy changes and often under-represent patient groups (for example, those with comorbidities). Furthermore, the continuous improvement approach of rapid learning can optimise interventions to the health needs of the patient populations served by different radiotherapy departments. The research activities have been organised into 7 interrelated work packages (WPs).
Work package 1: Determine the ethical, legal and regulatory acceptability of using rapid learning
Methods: Conceptual analysis of the legal, regulatory and ethical literature on trial consent will be carried out for determining the consent model in consultation with Citizens’ Juries. Qualitative interviews will be conducted to understand the practical effectiveness and patient experience of the developed model.
Work package 2: Implement statistical methods
Methods: Quality audit of data will be performed followed by the development of evaluation methods required for rapid learning. Interrupted time-series methods will be used to assess impact on patient outcomes over time.
Work package 3: Demonstrate clinical effectiveness of rapid learning to prospectively optimise heart dose limits
Methods: Acceptable toxicity profiles for radiotherapy and associated learning cycle-stopping criteria will be established using nominal group theory. Heart dose limit for all stage II-III patients - treated with radiotherapy - will be determined and refined further in the second and subsequent learning cycles.
Work package 4: Demonstrate economic evidence for the reduction of radiation dose compared with current practice
Methods: An individual patient-level, decision-analytic-model-based, cost-effectiveness analysis will be performed by using the expert opinion (for example, oncologists, radiotherapists, clinical scientists) combined with a systematic review of relevant existing decision analytic models. Furthermore, the degree of consensus about the use of the level 5 EQ-5D instrument as an outcome measure will be investigated using a mixed-method Delphi-survey
Work package 5: Investigate wider applicability and potential impact of data bias on the quality of evidence
Methods: In this work package, case studies of historic changes to radiotherapy practice will be identified through systematic qualitative content analysis. A minimum rapid-learning prototype dataset will be developed by combining with the results of quantitative analyses. Data quality will be assessed for each change using the audit approach developed in WP2, which will be used for producing statistical and imputation methods needed for analysis of emerging outcomes.
Work package 6: Assess feasibility of implementation across NHS
Methods: Healthcare professionals will be interviewed to gather an understanding of perceptions towards rapid learning and the practical experience of implementing it. In addition, interviews with individuals from key regulatory bodies, professional societies and opinion formers will be undertaken to understand the aspects of rapid learning that could potentially prevent widespread implementation. Data will be subjected to a framework approach for carrying out thematic analysis, taking a critical realist perspective.
Work package 7: Seek consensus for implementation in NHS radiotherapy centres
Methods: A wide range of traditional and innovative dissemination and communication activities will be used to raise awareness of rapid learning in radiotherapy among stakeholders. These will include workshops, publications, conferences, meetings, symposia, institutional promotions, through professional and clinical networks and digital approaches (social media, web).
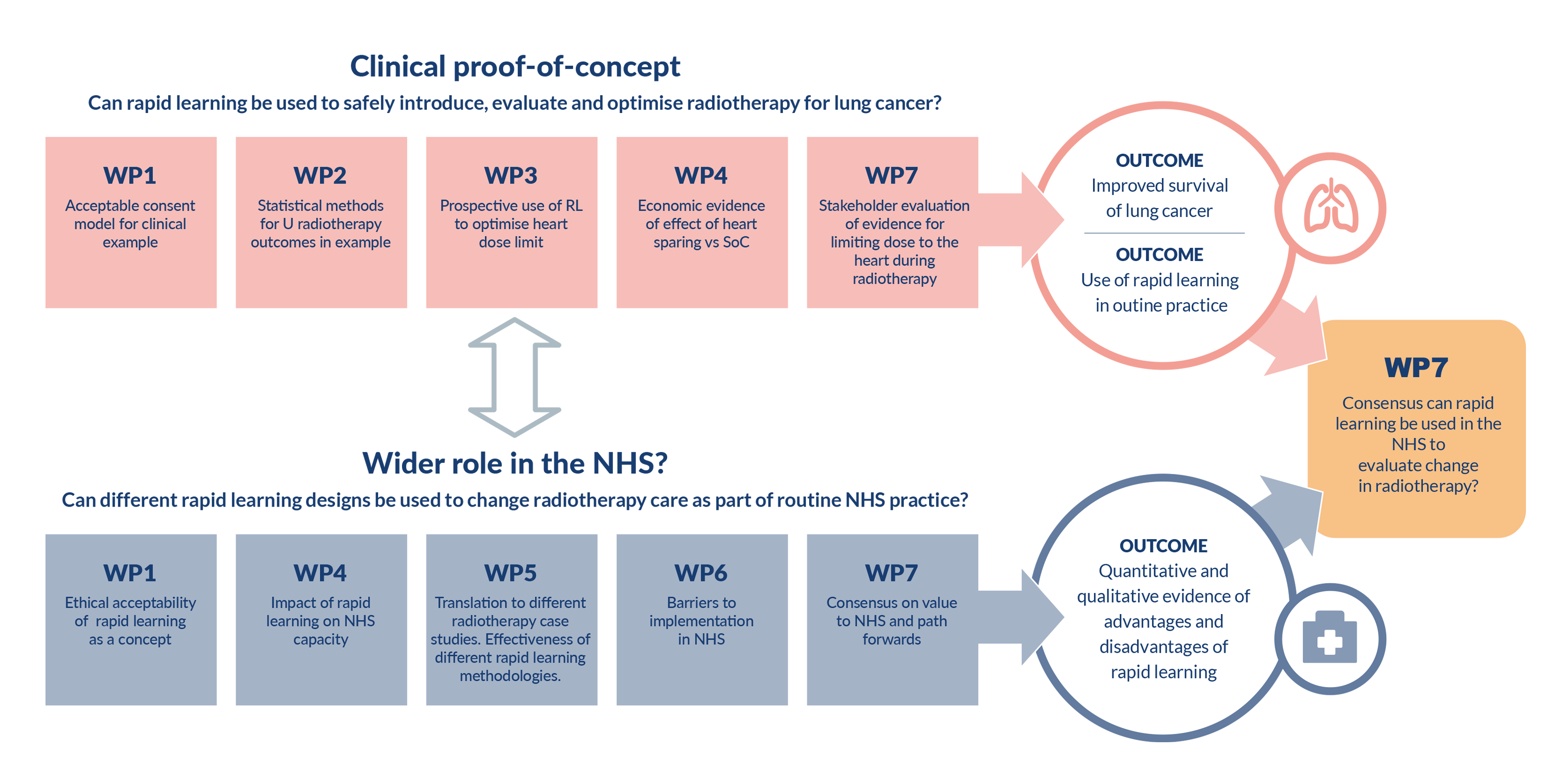
Figure 1: Programme work packages
Caption for Figure 1
To reflect the aims of the programme the work packages are conceptualised as two interrelated workstreams.
The first workstream entails assessing the clinical proof of concept, and asking the question: “Can rapid learning be used to safely introduce, evaluate and optimise radiotherapy for lung cancer?” The work is organised with the intention of achieving outcomes that improve survival of lung cancer and assess the use of rapid learning in routine practice.
This is done through the following activities:
- Work package 1 produces an acceptable consent model for the clinical example.
- Work package 2 produces statistical methods for radiotherapy outcomes in the example.
- Work package 3 looks at the prospective use of rapid learning to optimise the heart dose limit
- Work package 4 generates economic evidence of the effect of heart sparing versus Standard of Care (SoC)
- Work package 7 convenes a stakeholder evaluation of the evidence for limiting the dose to the heart during radiotherapy.
The second workstream assesses the wider role in the NHS and answers the question: “Can different rapid learning designs be used to change radiotherapy care as part of routine NHS practice?” The work is organised with the intention of achieving the outcome of obtaining quantitative and qualitative evidence of the advantages and disadvantages of rapid learning.
This is done through the following activities:
- Work package 1 looks at the ethical acceptability of rapid learning as a concept.
- Work package 4 determines the impact of rapid learning on NHS capacity.
- Work package 5 looks at the translation of different radiotherapy case studies and the effectiveness of different rapid learning methodologies.
- Work package 6 considers the barriers to implementation in the NHS.
- Work package 7 generates consensus on the value to the NHS and the path forwards.
Finally, the outputs of the 7 work packages and outcomes of the two work streams are consolidated in work package 7 to generate a consensus on whether rapid learning can be used in the NHS to evaluate change in radiotherapy.

