Published: 06 October 2023
Insulin delivered using smartphone technology helps pregnant women with type 1 diabetes manage blood sugars better. As a result, all pregnant women with the condition should be offered the technology. Those are the findings from a newly-published study, jointly funded by the NIHR and the Medical Research Council (MRC).
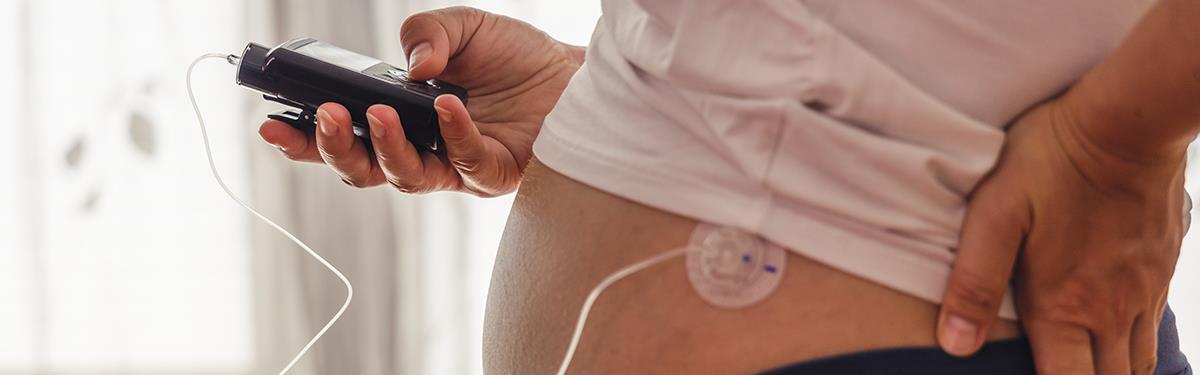
In the study, researchers trialled Hybrid Closed-Loop or Artificial Pancreas technology. This features an algorithm which sits on a smartphone. It communicates with the traditional continuous glucose monitoring and insulin pump systems.
The system adjusts insulin doses every 10-12 minutes according to blood sugar levels. This means it continuously responds to changes in blood sugar levels throughout pregnancy. The team compared this technology with the traditional continuous glucose monitoring and insulin systems. These see women supported by specialist diabetes maternity teams and require daily decisions to be made on insulin doses.
Widespread complications
Altered eating behaviours and hormonal changes during pregnancy can have a major impact. They mean most women struggle to reach recommended blood sugar targets. As a result, complications related to having type 1 diabetes are widespread during pregnancy. These complications can affect 1 in every 2 newborn babies. Risks can include:
- premature birth
- need for intensive care after birth
- being too large at birth, increasing the lifelong risk of overweight and obesity
Low blood sugars, excess weight gain, and high blood pressure during pregnancy are common symptoms.
The study involved 124 pregnant women with type 1 diabetes. They were aged 18-45 years and managed their condition with daily insulin therapy. Half were chosen to use the Hybrid Closed-Loop technology, the other half used traditional insulin pumps or multiple daily injection methods.
The women took part for approximately 24 weeks (from 10-12 weeks) until the end of pregnancy. The study took place in 9 NHS hospitals in England, Scotland, and Northern Ireland.
Consistent improvements
On average, pregnant women used the Hybrid Closed-Loop technology for more than 95% of the time. Using the technology helped to substantially reduce maternal blood sugars throughout pregnancy. Compared to traditional insulin therapy methods, women who used the technology spent more time in the target range for pregnancy blood sugar levels*. It was safely initiated during the first trimester, this is a crucially important time for babies’ development. The blood sugar levels improved consistently in mothers of all ages, regardless of their previous blood sugar levels, or previous insulin therapy.
These improvements were achieved without additional low blood glucose events. They were also achieved without additional insulin. Women using the technology gained 3.5 kg (equal to 7.7 lbs) less weight. They were also less likely to have blood pressure complications during pregnancy.
Importantly, women using the technology also had fewer antenatal clinic appointments and fewer out-of-hours calls with maternity clinic teams. This suggests the technology could save time for pregnant women and maternity services.
Professor Helen Murphy, Lead Author, University of East Anglia, said: “For a long time, there has been limited progress in improving blood sugars for women with type 1 diabetes, so we’re really excited that our study offers a new option to help pregnant women manage their diabetes.
“We know that for women with type 1 diabetes, unborn babies are exquisitely sensitive to small rises in blood sugars, so keeping blood sugar levels within the normal range during pregnancy is crucial to reduce risks for the mother and child. Previous studies have confirmed that every extra hour spent in the blood sugar target range reduces the risks of premature birth, being too large at birth and need for admission to neonatal intensive care unit. This technology is game changing, in that it will allow more women to have safer, healthier, more enjoyable pregnancies, with potential for lifelong benefits for their babies.”
The results have been published in The New England Journal of Medicine. The study was led by researchers at the University of East Anglia. Other contributors included:
- Norfolk and Norwich University Hospitals NHS Foundation Trust
- Cambridge University Hospitals NHS Foundation Trust
- the Wellcome-MRC Institute of Metabolic Science, University of Cambridge
- Leeds Institute of Cardiovascular and Metabolic Medicine, University of Leeds
- the Institute of Cardiovascular and Medical Sciences, University of Glasgow
- King’s College Hospital NHS Foundation Trust
- the Regional Centre for Endocrinology and Diabetes, Royal Victoria Hospital, Belfast
- Barnard Health Research
- the Usher Institute
- the Centre for Cardiovascular Science, University of Edinburgh
- the Jaeb Center for Health Research
This study was funded by the Efficacy and Mechanism Evaluation (EME) Programme. The EME Programme is a partnership between NIHR and the MRC. For more information on this study, please visit our Funding and Awards page.
*(68% vs 56% – equivalent to an additional 2.5-3 hours every day throughout pregnancy)

