Published: 13 October 2020
Researchers today have called on more volunteers from Black, Asian and Minority Ethnic backgrounds and the over 65s to participate in vital COVID-19 clinical studies to ensure that any vaccines developed will work for as many people as possible.
Ethnic minorities are currently underrepresented in vaccine clinical trials taking place across the UK, with only 11,000 volunteers from Asian and British Asian backgrounds while 1,200 are Black, African, Caribbean or Black British.
This comes as the number of people in the UK who have signed up to take part in COVID-19 research through the NIHR and NHS Digital delivered NHS Covid-19 vaccine research registry hits 270,000, just six months after the first studies opened for recruitment.
With 93% people from non-ethnic minority groups having signed up, thousands of people from different ages and backgrounds are urgently needed to help speed up their development and ensure they work effectively for the whole population.
Black, Asian and Minority Ethnic communities are disproportionately affected by Covid-19, with this group at greater risk of becoming very sick with COVID-19 than the white population in the UK.
NIHR researchers have found patients of Black ethnicity have an increased risk of requiring hospital admission for COVID-19, while patients of Asian ethnicity have an increased risk of dying in hospital from COVID-19. Black men and women in particular are nearly twice as likely as white people to die from COVID-19.
Large-scale clinical studies with a diverse range of volunteers will aid researchers to better understand the effectiveness of each vaccine candidate. There are currently six different Covid-19 vaccines currently progressing in the UK, including US biotech company Novavax at a number of NIHR regional sites across the UK in Lancashire, the Midlands, Greater Manchester, London, Glasgow and Belfast.
UK researchers are also encouraging additional volunteers from people with underlying health conditions such as chronic diseases and the over 65s, to be part of clinical trials so that any vaccines developed will work for as many people as possible.
The NHS Vaccine Registry was launched in July to help create a database of people who consent to be contacted by the NHS to take part in clinical studies with the aim to help speed up the development of a safe and effective vaccine against coronavirus.
The Registry has been developed as part of the UK Government’s Vaccine Taskforce, in partnership with the NIHR, NHS Digital, and the Northern Ireland, Scottish and Welsh Governments and the Northern Ireland Executive.
Divya Chadha Manek, Head of Business Development for the NIHR Clinical Research Network and Clinical Trials Workstream Lead at UK Vaccines Taskforce said:
“NIHR-supported research has clearly demonstrated evidence that people from Black and Asian ethnic groups are disproportionately affected by COVID-19. It is imperative that more people from Black, Asian and different groups sign up to the NHS Registry as soon as possible, so we can ensure COVID 19 vaccines are effective for as many people as possible.
"We need people from all our communities to play their part by taking part in COVID 19 vaccine research. Essentially, we need to help ourselves by addressing the challenge of the pandemic through research."
Chair of the Government’s Vaccine Taskforce, Kate Bingham said:
“The only way to check how well a coronavirus vaccine works is to carry out large-scale clinical trials involving thousands of people. Researchers need data from different communities and different people to improve understanding of the vaccines. The only way to get this is through large clinical trials.
“We want to ensure the data we get actually represents the different people from different backgrounds in the UK. This includes people who are over 65, frontline healthcare workers, or have existing health conditions, and we need people from the communities which have been disproportionately affected by the pandemic from black, Asian and other minority ethnic backgrounds.”
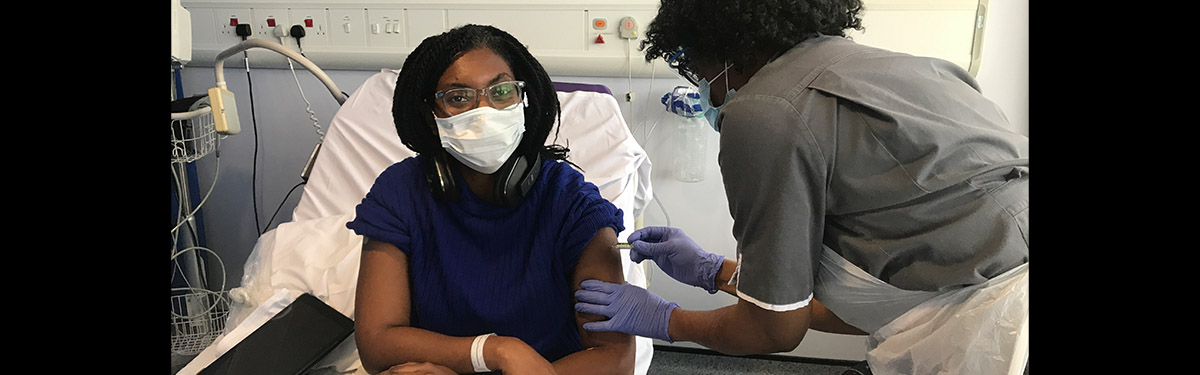
Minister for Equalities Kemi Badenoch, who is volunteering for Covid-19 vaccine trials being conducted by US biotechnology company Novavax at Guy’s and St Thomas’ NHS Foundation Trust, London, said:
“The UK is leading the world in the search for a Covid-19 vaccine. At home, we have to ensure every community trusts a future vaccine to be safe and that it works across the entire population.
“But with less than half a per cent of people on the NHS Vaccine Registry from a Black background, we have a lot more work to do.
“That is why I am urging more people from the ethnic minority backgrounds to join me in signing up to the NHS Vaccine Registry and taking part in a trial. Together we can be part of the national effort to end this pandemic for good.”
Business Secretary Alok Sharma said:
“Coronavirus can affect anyone regardless of their background, age or race. To ensure we can find a safe and effective vaccine that works for everyone, we all need to get involved.
“That’s why we are urging more people to support our incredible scientists and join the 270,000 people who have already signed-up so we can speed up efforts to find a vaccine to defeat this virus once and for all.”
Further information
Black, Asian and minority ethnic participation in vaccine research is explored in detail in the latest podcast in the series, COVID-19: the search for a vaccine.
The UK public can support the national effort to speed up vaccine research and receive more information about volunteering for clinical studies by visiting www.nhs.uk/researchcontact.
Visit the Be Part of Research website for more information about taking part in research and other opportunities to take part in COVID-19 research.

